A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton.
- Francis Sutton
- Date:
- 1890
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton. Source: Wellcome Collection.
486/592 page 470
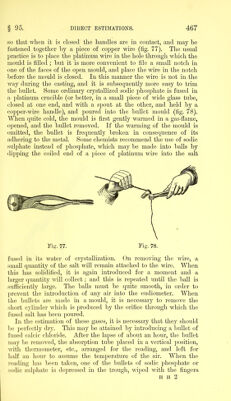
![After absorption of carbonic anhydride by bullet of potassic hydrate. Gas Dry. Height of mercury in trough . = 172*0 m.m. Height of mercury in absorption eudio- meter . . . = 62*5 m.m. Column of mercury in eudiometer = b = 109*5 m.m. Height of mercury in eudiometer Correction for error of meniscus 62*5 m.m. 0*8 m.m. 63*3 m.m. Volume in table corresponding to m.m. Temperature . Barometer 63*3 = V = 69*35 = t = 10*8° = B =766*0 m.m. V x (B — b) v “ 1000 X (1 + 0*003665#) 69*35 x (766*0- 109*5) 1000 x [1 + (0*003665 x 10*8)] 69*35x 656*5 1000 x 1 *039582 43w 95 log. 69*35= 1*84105 log. 656*5 =2*81723 4*65828 log. (1000 x 1*039582)= 3*01686 1*64142 = log. 43*795 = V1 Corrected volume of air = 43*795 Air + CO2 =61*994 Air =43*795 C02= 18*199 61*994 18*199 : : 100 18*199x100 'x~ 61-995 : x = percentage of CO2 29*355 Percentage of CO2 in mixture of air and gas = 29*355.](https://iiif.wellcomecollection.org/image/b28127006_0488.jp2/full/800%2C/0/default.jpg)