The periodic classification and the problem of chemical evolution / by George Rudorf.
- Rudorf, George.
- Date:
- 1900
Licence: In copyright
Credit: The periodic classification and the problem of chemical evolution / by George Rudorf. Source: Wellcome Collection.
83/252 page 63
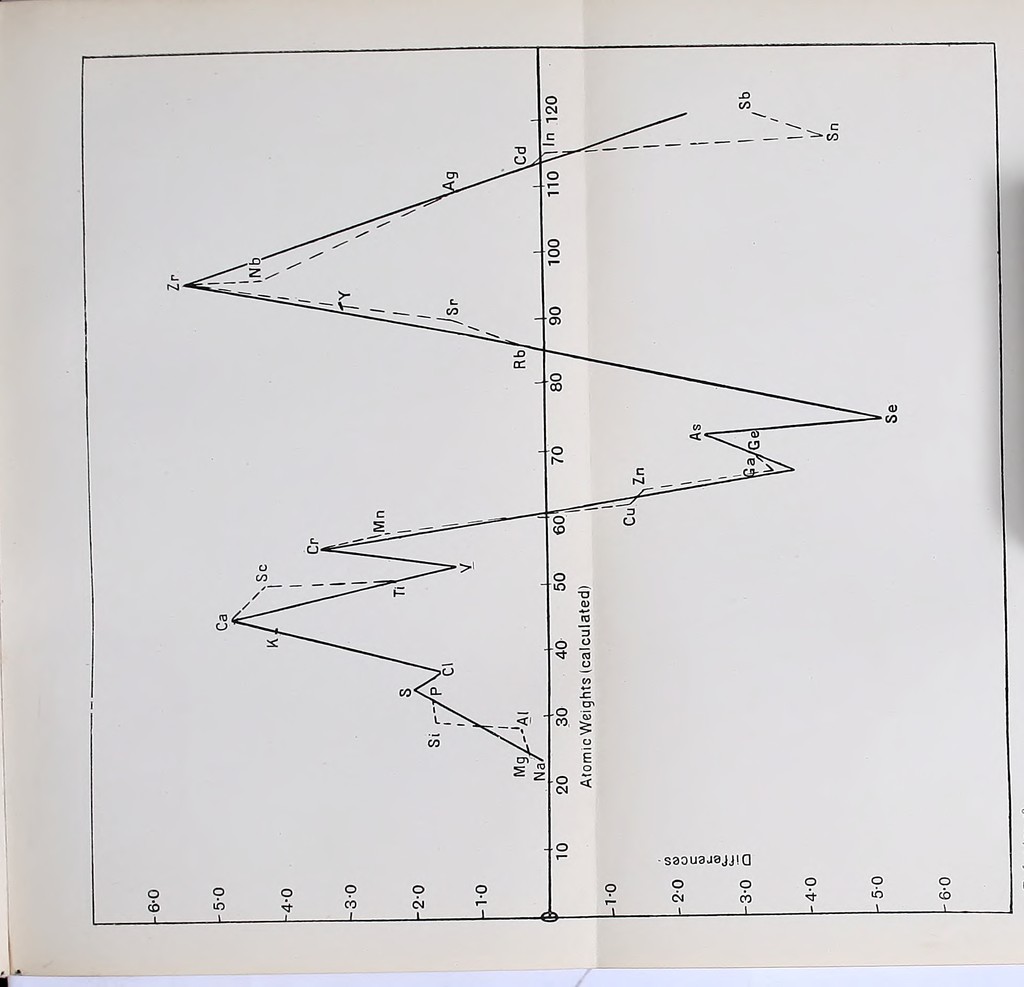
![Cd. In. Sn. Sb. Te. I. Calculated . 110-88 115-71 117-14 123-27 123-64 ? Found 112-38 113-85 119-05 120-43 127-49 126-85 Difference . -1-50 + 1-86 -1-91 + 2-84 -3-85 ? Ba. La. Ce. Hg. Tl. Pb. Calculated . 135-76 138-39 140-33 200-69 203-46 206-13 Found 137-43 138-64 139-35 200-00 204-15 206-92 Difference . -1-67 -0-25 + 0-98 -0-69 -0-69 -0-79 Here again the results are too irregular to lead to any definite law. The differences are in most cases small, except for antimony and tellurium. Various attempts have from time to time been made to reduce the numbers expressing the atomic weights to order. o De Chancourtois.—Perhaps the first attempt of the kind may be ascribed to De Chancourtois in 1862, who represented the elements on a helix described round a right circular cylinder, and having a constant slope of 45° to the axis. This has been described and figured in Chap. I. (q.v.). His results do not lead to much, and, being himself a geologist and a mathe- matician, he was not able to follow up his work from a chemical standpoint. Mills.—In 1884 Dr E. J. Mills published a paper on the “Numerics of the Elements”1 and another2 in 188G. He arranges all the elements in order of their atomic weights, and divides them into sixteen groups. He then finds that the atomic weights can be em- bodied in an expression of the form i/ = K-B/3% where y represents the atomic weight and x is an integer different for different elements, Iv is nearly 1 Phil. Mag. [5], 18, p. 393. 2 Phil. Mag. [5], 21, 151,](https://iiif.wellcomecollection.org/image/b28105102_0083.jp2/full/800%2C/0/default.jpg)