Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt.
- International Congress on Isozymes
- Date:
- [cl983]
Licence: Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0)
Credit: Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt. Source: Wellcome Collection.
34/256 page 14
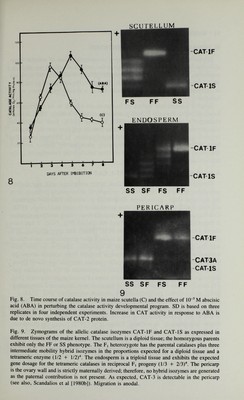
![14 / Scandalíos III. INTRACELLULAR COMPARTMENTATION Higher organisms typically display a compartmentation of cellular func¬ tions that is of fundamental importance to the process of cellular metabolism, regulation, and differentiation. Compartmentation provides alternative means for the catalysis of individual reactions. Therefore, the location of an enzyme, or isozyme, at the correct intracellular site is a crucial requirement of its function. A great number and variety of isozymes are known to be differentially localized within the cells of both plants and animals [Gottlieb, 1982; Schnar- renberger et al, 1983; Weeden, 1983; Masters, 1983]. Such intracellular compartmentation is very frequently tissue specific and time specific. Usually, organelles are isolated by differential centrifugation, as different organelles have different densities, and subsequently identified by marker enzymes specific to a given organelle and by electron microscopic examination of the appropriate subcellular fractions. A. Intracellular Localization of CAT-1, CAT-2, and CAT-3 During kernel development, catalase is not associated with any distinct subcellular organelles. However, as sporophytic development proceeds from the dry seed stage, glyoxysome biogenesis in scutella becomes apparent, reaching a peak at the fourth day after germination [Longo and Longo, 1970]. Cell-fractionation studies show that in scutella from young seedlings catalase is associated with the glyoxysomes and the cytosol [Fig. 10А]. It has been shown [Scandalios, 1974; Scandalios et al, 1980b] that approximately 50% of the total catalase present in scutella at day 4 after germination is associated with the glyoxy somes; the remainder is cytosolic. There is no detectable catalase associated with the mitochondria isolated from scutella at any de¬ velopmental stage. There is no particular catalase isozyme specific to glyox- ysomes [Scandalios, 1974]; the isozyme complement of the organelles par¬ allels that characteristic of the particular stage of scutellar development [Fig. 11; see also insert Fig. 14]. Purity of fractions was determined enzymolog- ically (using marker enzymes) and by electron microscopy (Fig. 12A). lodination experiments using lactoperoxidase clearly indicate that the or- ganelle-associated catalase is inside the glyoxysome structure and not merely bound to the outside surface of the organelles [P.H. Quail and J.G. Scan¬ dalios, unpublished]. In addition. Longo et al [1972], using the diaminob- enzidine (DAB) histochemical reaction, demonstrated that the membranes of maize glyoxysomes become heavily stained after incubation with DAB and H2O2. Their experiments also suggested that catalase may be present in the matrix as well as being membrane bound. The possible role of catalase in the function of glyoxysomes in converting fatty acids to sugars has been discussed elsewhere [Cooper and Beevers, 1969; Tolbert, 1971].](https://iiif.wellcomecollection.org/image/b18019742_0035.JP2/full/800%2C/0/default.jpg)