Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt.
- International Congress on Isozymes
- Date:
- [cl983]
Licence: Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0)
Credit: Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt. Source: Wellcome Collection.
44/256 page 24
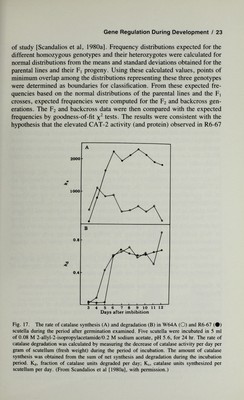
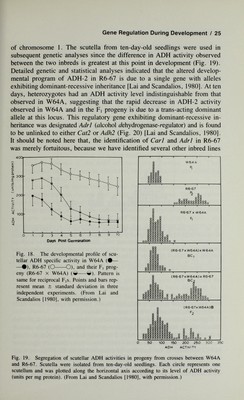
![24 / Scandalios is due to a single locus with additive alleles. By using electrophoretic variants of CAT-2 in genetic crosses, it was determined that the regulatory locus is loosely linked to Cat2 on the short arm of chromosome 1, approximately 37 map units away. This trans-acting locus has been designated Carl (catalase- regulator). Although turnover studies [Scandalios et al, 1980a] indicate that Carl acts by regulating the rate of CAT-2 synthesis, the precise mode of action or level of control of Carl over Cat2 is not yet understood. As a trans-acting locus, Carl must somehow transmit a molecular signal to the Cat2 locus or to its nascent mRNA to influence its expression. If so, it can be inferred that there must be specific receptors for these signals to ensure that they act on the correct locus (or its transcript), and there must be DNA sequences that define their site of action. An alternate model might be that, at approximately four days after germination, the Carl gene in W64A re¬ sponds to a signal to decrease CAT-2 synthesis in the scutellum, while in R6-67, the Carl gene does not respond to this signal and results in an elevated steady-state level of CAT-2 protein. The chemical nature of temporal gene signals remains an open question. To account for the specificity of such signals, one must assume that they are of a macromolecular nature (ie, protein, RNA, DNA). Whatever the nature of the signalling molecules may be, the primary question is the molecular mechanism by which the necessary information is encoded to instruct tem¬ poral genes as to where and when to function. It has been proposed [Paigen, 1979; Soil, 1979] that the existence of temporal genetic elements may suggest that developing eukaryotic organisms possess something equivalent to a clock in their nuclei with a mechanism for telling time, along with some pro¬ grammed mechanism for informing a given copy of a gene of its spatial location. Carl provides the first evidence in higher plants that a temporal gene can specifically program the level of a single species of an enzyme encoded in a distant structural gene {Cat2) on the same chromosome. B. Genetic Regulation of the Alcohol Dehydrogenase Developmental Program Alcohol dehydrogenase (ADH) activity in the scutellum of most maize inbred lines (eg, W64A) normally decreases following seed imbibition [Scan¬ dalios and Felder, 1971]. In line R6-67, ADH activity is unusually high in the scutellum of the dry seed and remains at a level nearly two to three times that observed in line W64A during the ten days following seed imbibition (Fig. 18). Using rocket Immunoelectrophoresis to quantitate the amount of ADH protein, it was determined that this variation in ADH activity is pri¬ marily due to changes in the amount of ADH-2 protein, the major isozymic form of maize ADH which is encoded in a structural gene on the long arm](https://iiif.wellcomecollection.org/image/b18019742_0045.JP2/full/800%2C/0/default.jpg)