Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt.
- International Congress on Isozymes
- Date:
- [cl983]
Licence: Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0)
Credit: Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt. Source: Wellcome Collection.
56/256 page 36
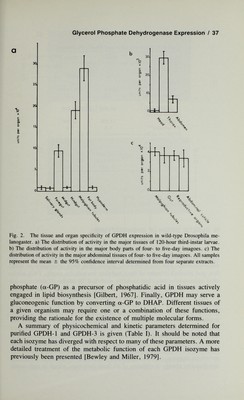
![36 / Bewley This period also marks a switch in the pattern of isozyme expression where GPDH-1 and GPDH-2 appear. This pattern of isozyme expression is retained throughout the remainder of the adult life cycle. The developmental expression of GPDH-specific CRM in a wild inbred laboratory strain of Drosophila is illustrated (Fig. lb). The CRM level in 48-hour-old adults (rocket area = 166 mm^) is about three times the level found in 120-hour third instar larvae (rocket area = 57 mm^). In addition, the relative CRM level in each developmental stage is nearly equivalent to the changes seen in total GPDH activity of Figure la. Since mixing exper¬ iments of crude extracts from each developmental stage give no evidence for endogenous inhibitors or activators, these results likely reflect differential rates of enzyme accumulation with respect to the time-specific expression of the structural gene, Gpdh^. More specifically, it has recently been dem¬ onstrated that the developmental expression is largely the result of changing rates of enzyme synthesis (D. Sullivan, personal communication). Each isozyme is also distinct with respect to tissue-specific expression. The majority of GPDH-3 activity in larvae is localized in the fat body, a tissue metabolically analogous to mammalian liver and the major site of lipid metabolism and storage in insects [Kilby, 1963], and Malpighian tubules (Fig. 2a). The remainder of GPDH-3 activity is ubiquitously distributed among the other larval tissues in low amounts. In the imago, the majority of the activity is associated with the thorax, where GPDH-1 is the predominant isozymic form (Fig. 2b). All three isozymes are found in limited amounts in the head tissue, while GPDH-3 is the major form found in the abdomen of the imago [Wright and Shaw, 1969; Rechsteiner, 1970], where it is ubiq¬ uitously distributed among the major tissues (Fig. 2c). GPDH-1 is associated with a tissue unique to the imago, thoracic flight muscle, and plays an important role in the metabolism of insect flight [Sacktor, 1965]. III. METABOLIC DIFFERENTIATION The reaction catalyzed by GPDH (DHAP + NADH + H^ ^ a-GP + NAD^) suggests that it serves a central role in either carbohydrate or lipid metabolism, or in the coupling of these two processes. Depending on the function of the tissue within which GPDH resides, at least four metabolic roles can be envisioned. First, it can serve to maintain the proper NAD*^ levels in tissues with high glycolytic capacity [White and Kaplan, 1969]. Second, it can function to provide reducing equivalents to the electron trans¬ port chain by its participation in the a-glycerol-phosphate cycle [Sacktor, 1965]. Third, GPDH may play an important role in providing a-glycerol- ! /](https://iiif.wellcomecollection.org/image/b18019742_0057.JP2/full/800%2C/0/default.jpg)