Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt.
- International Congress on Isozymes
- Date:
- [cl983]
Licence: Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0)
Credit: Gene expression and development : the third of five volumes constituting the proceedings of the 4th International Congress on Isozymes, held in Austin, Texas, June 14-19, 1982 / editors, Mario C. Rattazzi, John G. Scandalios, Gregory S. Whitt. Source: Wellcome Collection.
71/256 page 51
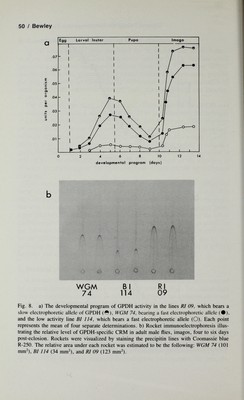
![Glycerol Phosphate Dehydrogenase Expression / 51 tained for enzyme activity and indicates that the enzyme activity differences observed between lines must be due to differential rates of enzyme accu¬ mulation throughout development (Fig. 8b). The enzyme has been purified from each of the strains illustrated in Figure 8, and all three variants have similar kinetic, immunological, and physicochemical parameters except for the pi differences associated with the electrophoretic differences observed (line Rl 09 = pi 5.9; WGM 74 and BI114 = pi 6.1). These results suggest that the site causing a constant threefold difference in activity throughout development is not associated with any qualitative change in enzyme struc¬ ture. We have developed a microinjection technique for radiolabeling Droso- phila proteins in vivo and have subsequently used this technique in combi¬ nation with immunological precipitation of GPDH to examine turnover pa¬ rameters of GPDH in these three variant lines [Wilkins et al, 1982; Shaffer et al, 1982]. Short-term uptake studies that follow the incorporation of ^H- leucine into de novo synthesized immunoprecipitable GPDH are illustrated in Figure 9a. It is clear that GPDH is rapidly labeled in each line, that the incoфoration of^H-leucine follows the same time course regardless of ge¬ notype, and that the maximum specific radioactivity (P*max,CPM/mg wet weight) was reached simultaneously for each line. The striking difference between variant lines is seen in the absolute p*niax values attained. From the data, it is clear that within the four-hour time period of the experiment, lines R109 and WGM 74 incorporated 3.05 and 2.76 times more label, respectively, into immunoprecipitable GPDH than did the low activity line Bl 114 (Fig. 10a). The kinetics of ^H-leucine loss from immunoprecipitable GPDH in lines Rl 09, WGM 74, and Bl 114 are illustrated (Fig. 9b). The rate of loss of label between the three lines is nearly identical and exhibits typical first- order rate kinetics expected for in vivo intracellular protein degradation. It is therefore apparent that the observed differences in steady-state levels of GPDH-specific CRM in these three lines can only be accounted for by dif¬ ferential rates of GPDH synthesis. In addition, the rates of ^H-leucine in- coфoration and loss in total TCA precipitable protein is similar in all three lines, indicating that differential rates of total protein turnover cannot account for the line-specific differences observed for GPDH turnover parameters. Genetic analysis of the site affecting the rate of GPDH synthesis is sum¬ marized in Figure 10. The site segregates as a single genetic element with additive inheritance. In addition, this site has been demonstrated to lie within the boundaries of the deficiency Df(2L)GdhA, with breakpoints at 25E1-F and 26B1-E1, which includes the structural element Gpdh^. This evidence, in addition to the fact that the site co-segregates with electrophoretic markers of the structural element (Fig. 10a), indicates that there is tight linkage with the structural element and that the two elements may be contiguous. Further,](https://iiif.wellcomecollection.org/image/b18019742_0072.JP2/full/800%2C/0/default.jpg)