A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton.
- Francis Sutton
- Date:
- 1882
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton. Source: Wellcome Collection.
Provider: This material has been provided by University of Bristol Library. The original may be consulted at University of Bristol Library.
436/508 page 420
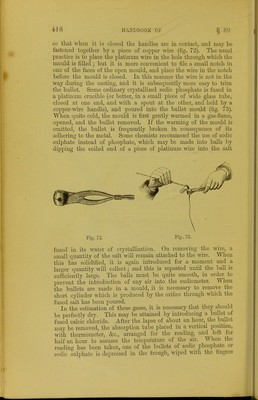
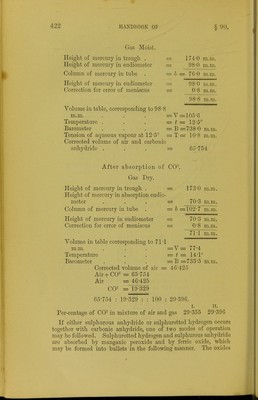
![marked with the nail wlien cold. Before use the balls must be slightly moistened with water; and if large quantities of gas have to be absorbed, the bullet must be removed after some hours, washed with water, and returned to the absorption tube. The absorption may extend over twelve or eighteen hours. In order to ascertain if it is completed, the potash ball is removed, washed, again introduced, and allowed to remain in contact Avith the gas for about an hour. If no diminution of volume is observed the operation is finished. The following analyses of a mixture of air and carbonic anhydride will serve to show the mode of recording the observa- tions and the methods of calculation required. Analysis of a Mixture of Air and Carbonic Anhydride. 1. Gas Saturated with Moisture. Height of mercury in trough . = 171'8 ni.nj. Height of mercury in absorption eudio- meter . . . = S9'0 m.m. Column of mercury in tube, to be sub- tracted from the height of barometer= h ■= 82'8 m.m. Height of mercury in eudiometer = 89*0 ra.m. Correction for error of meniscus = 0*8 m.m. 89-8 m.m. Volume in table corresponding to 89 -8 m.m. . . . =V= 96-4 Temperature at which the reading was made . . . =t= 12-2° Height of barometer at time of obser- vation . . . =B=76525 m.m. Tension of aqueous vapour at 12-2° =T= 10*6 m.m. y X (B-Z;-T) 1000 X (l + 0-00366.5^)~ 9G4 X (765-25-82-8-10-6) _ 1000 X [1 (0-003665 x 12-2)] 96-4 X 671-85 1000 X 1-044713 ~ log. 96-4 = 1-98408 log. 671-85 = 2-82727 4-81135 log. (1000 X 1-044713) = 3-01900 1-79235 = log. 61-994 = V» Corrected volume of air and CO- = = 61-994.](https://iiif.wellcomecollection.org/image/b21443658_0436.jp2/full/800%2C/0/default.jpg)