A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton.
- Francis Sutton
- Date:
- 1882
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton. Source: Wellcome Collection.
Provider: This material has been provided by University of Bristol Library. The original may be consulted at University of Bristol Library.
468/508 page 452
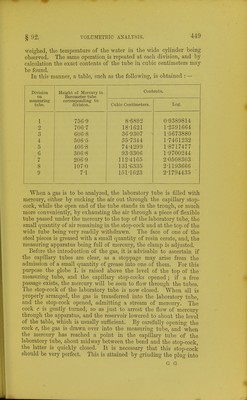
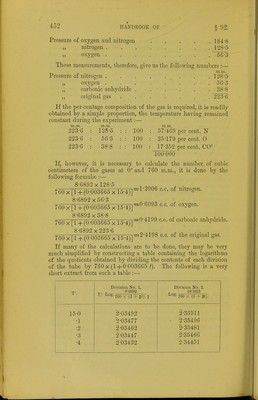
![Pressure of oxygen and nitrogen nitrogen . it oxygen 184-8 128-5 56-3 These measurements, therefore, give us the following numbers Pressure of nitrogen .... oxygen . . _ . carljonic anhydride . original gas . . • . If the per-centage composition of the gas is required, it is readily obtained by a simple proportion, the temperature having remained constant during the experiment:— an.m. 128-5 5G-3 38-8 223-G m.m. 223-G m.m. 12S-5 : : 100 223-6 56-3 : : 100 223-6 38-8 : : 100 m.m. 57-469 per cent. N 25-179 per cent. 0 17-352 per cent. CO- 100-000 If, however, it is necessary to calculate the number, of cubic centimeters of the gases at 0° and 760 m.m., it is done by the following formulas:— 8-6892 x 128-5 ^ . T=1*3906 c.c. of nitrogen. 760 x[l +(0-003665x 15-4)] 8-6892 X 56-3 760 x[l +(0-003665 x 15-4)] 8-6892x38-8 760 x[l+(0-003665 X 15-4)] 8-6892x223-6 =0-6093 c.c. of oxygen. =0-4199 c.c. of carbonic anhydride. :2*4198 c.c. of the original gas. 760 x[l+(0 003665 X 15-4)] If many of the calculations are to be done, they may be very much simplified by constructing a table containing the logarithms of the quotients obtained by dividing the contents of eacb division of the tube by 760 x (1+ 0-003665 /). The following is a very short extract from such a table :— 15-0 •1 •2 •3 •4 Division No. X. ^. ^ 8-6892 1\: Log. •760 X (1 + gt;. I 2-03492 2-03477 2-03462 2-03447 2-03432 Division No. 2. 18-1621 Log. ■ 760 X (1 + 5t). 2-35511 2-35496 2-35481 2-35466 2-34451](https://iiif.wellcomecollection.org/image/b21443658_0468.jp2/full/800%2C/0/default.jpg)