A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton.
- Francis Sutton
- Date:
- 1882
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids, and gases : adapted to the requirements of pure chemical research, pathological chemistry, pharmacy, metallurgy, manufacturing chemistry, photography, etc., and for the valuation of substances used in commerce, agriculture and the arts / by Francis Sutton. Source: Wellcome Collection.
Provider: This material has been provided by University of Bristol Library. The original may be consulted at University of Bristol Library.
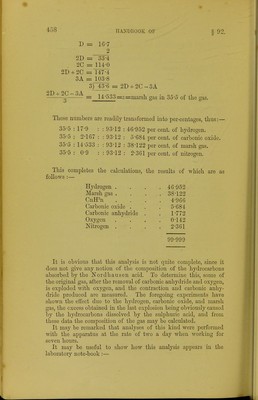
471/508 page 455
![The gas is again passed into the laboratory tube, and a coke ball soaked in fuming sulpliuric acid, left in contact with the gas for an hour; the bullet is then withdrawn, and some potassic hydrate introduced and left in the tube for ten minutes, in order to remove the vapours of sulphuric anhydride, and the sulphurous and carbonic anhydrides formed during the action of the Ifordhausen acid on the gas. The gas is now measured again. Height of mercu]'y in barometer tube . . 969'3 „ ,, before absorbing CnlPn 983-3 „ after . ■ 969-3 Pressure of CnWn M'O The per-centage of these hydrocarbons is thus formed :— Gas containing CnlPn. H. CO. CH^. CnH2n. 276-5 : U-0 : : 98-086 : 4-966 per cent. CnH'^n It now remains to determine the hydrogen, carbonic oxide, marsh gas, and nitrogen in a portion of the residual gas. The laboratory tube is therefore removed, some of the gas allowed to escape, and another laboratory tube adapted to the apparatus. The portion of g-s remaining is expanded to a lower ring (in this special case to the third division), and the tension measured :— Height of mercury in the barometer tube . . 642-2 „ measuring tube . . 606-7 Pressure of residue 35-5 An excess of oxygen has now to be added. Por this purpose the gas is passed into the laboratory tube, and about five times its volume of oxygen introduced from a test tube or gas pipette. The necessary quantity of oxygen is conveniently estimated by the aid of rough graduations on the laboratory tube, which are made by introducing successive quantities of air from a small tube in the manner previously described for the calibration of the eudiometei-s. After the introduction of the oxygen, the mixed gases are passed . into the eudiometer and measured. Height of mercury in the eudiometer after addition of 0 789-5 The mixture has now to be exploded, and when the pressure is considerable, it is advisable to expand the gas so as to moderate the violence of the explosion. When suihciently dilated, the stop-cock at the bottom of the eudiometer is closed, tlie level of the water lowered beneath the platinum wires by depressing the syphon, and the spark passed. The explosion should be so powerful that it should be audible, and the tiash visible in not too bright daylight.](https://iiif.wellcomecollection.org/image/b21443658_0471.jp2/full/800%2C/0/default.jpg)