The absorption spectra of some substituted pyrazines and their salts / by Frank Tutin and Frederic W. Caton.
- Tutin, Frank.
- Date:
- 1910
Licence: Attribution 4.0 International (CC BY 4.0)
Credit: The absorption spectra of some substituted pyrazines and their salts / by Frank Tutin and Frederic W. Caton. Source: Wellcome Collection.
Provider: This material has been provided by The Royal College of Surgeons of England. The original may be consulted at The Royal College of Surgeons of England.
11/14 page 2532
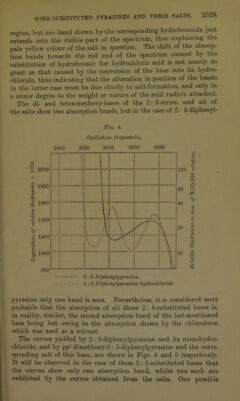
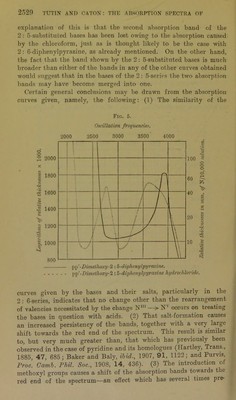
![derivative of the 2: 6-substituted base, and, when dissolved in chloroform, exhibits an intense green fluorescence. pp7-Dimethoxy-2 : 5-diphenylpyrazine monohydrobromide, C4H2N2(C6H4-OMe)2,HBr, was prepared in a manner similar to the salt last described. It formed orange-coloured needles: 0'2781 gave 0T373 AgBr. HBr = 21'3. C]8Hlc02N2,HBr requires HBr = 21'7 per cent. pp7-Dimethoxy-2: 5-diphenylpyrazine monohydrobromide, like the corresponding hydrochloride, is fluorescent in chloroform solution. pp7-Dimethoxy-2 : 5-diphenylpyrazine monosulphate, C4H2N2(C6H4-0Me)2,H2S04, separated in yellow needles on adding sulphuric acid to a warm solution of the respective base in glacial acetic acid, and cooling the mixture. It dissociates readily, and, like the last-mentioned two salts, is fluorescent in chloroform solution: 0-2700 gave 01581 BaS04. H2S04 = 24’6. C18H1602N2,H2S04 requires H2S04 = 25‘1 per cent. When to a solution of pp7-dimethoxy-2: 5-diphenylpyrazine in chloroform, concentrated sulphuric acid was added, a yellow solution of the monosulphate was first formed, which ex- hibited a brilliant green fluorescence, but on introducing an excess of the acid the base was dissolved by the latter, yielding an intensely violet-coloured liquid below the chloroform. Sufficient ethyl acetate was then added to render the mixture homogeneous, and the liquid kept a few hours, when pp1-dimethoxy-2 : 5-diphenyl- pyrazine disulphate, C4H2N2(C6H40Me)2,2H2S04, separated in small prisms, resembling in colour crystals of potassium per- manganate. This disulphate is very unstable; it dissociates in ordinary air, yielding the yellow monosulphate, and, if the air be unusually damp, complete dissociation ensues. The dissociated mixture may, however, be successively reconverted into the yellow monosulphato and the deep violet-coloured disulphate by desic- cation : 0T224 gave 0-1200 BaS04. H2S04 = 41T. C18H10O2N2,2H2SO4 requires H2SO4 = 40'2 per cent. This disulphate dissociated so readily that it could not be washed with any solvent, and it is owing to this fact that the analysis indicated a somewhat high percentage of sulphuric acid. Salts of pp'-Dimethoxy-2 : 6-diphenylpyrazine.—This base readily yielded a crystalline monoliydi-ochloride, monohydrobromide, and monosulphate, the first of which has been described in the preceding](https://iiif.wellcomecollection.org/image/b22433181_0013.jp2/full/800%2C/0/default.jpg)