The absorption spectra of some substituted pyrazines and their salts / by Frank Tutin and Frederic W. Caton.
- Tutin, Frank.
- Date:
- 1910
Licence: Attribution 4.0 International (CC BY 4.0)
Credit: The absorption spectra of some substituted pyrazines and their salts / by Frank Tutin and Frederic W. Caton. Source: Wellcome Collection.
Provider: This material has been provided by The Royal College of Surgeons of England. The original may be consulted at The Royal College of Surgeons of England.
12/14 page 2533
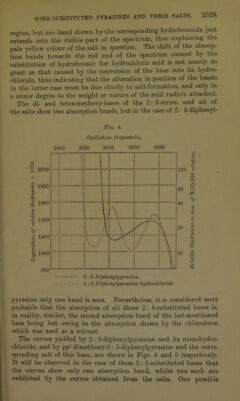
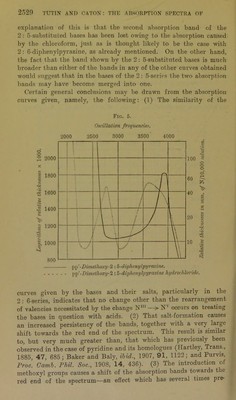
![paper (p. 2506). No indication of the formation of a dihydro chloride could be obtained, but the dihydrobromide was obtained as a very deep violet-coloured solid by passing dry hydrogen bromide over the solid base. A deep violebcoloured solution of the disulphate in concentrated sulphuric acid was formed, but this salt could not be crystallised. pp'-Dimethoxy-2 : 6-diphenylpyrazine monohydrobromide, C1H2N2(C6H4-OMe)2,HBr, crystallised in deep yellow needles on the addition of a little con- centrated hydrobromic acid to a solution of the base in a mixture of ethyl acetate and alcohol: 0'1685 gave 0-0832 AgBr. HBr = 21-3. C18HI602N2,HBr requires HBr = 21-7 per cent. This salt is not fluorescent, thus differing from its 2: 5-substituted isomeride. pp'-Dimethoxy-2 : 6-diphenylpyratine monosulphate, C4H2N2(C6H4*0Me)2,H2S04, was obtained on adding concentrated sulphuric acid to a solution of the respective base in ethyl acetate. It was not fluorescent, and formed pale yellow needles, which dissociated fairly readily: 0 2635 gave 01549 BaS04. H2S04 = 24'7. C]8Hi602N2,H2S04 requires H2S04 = 25'1 per cent. Salts of mm'pp'-Tetramethoxy-2 : 5-diphenylpyrazine. — The amount of this base available was very small, and therefore only its behaviour towards sulphuric acid was investigated. It was markedly more basic than the previously mentioned bases. When treated with a small amount of sulphuric acid in glacial acetic acid solution, it yielded mm'pp'-tetramethoxy-2: 5-diphenylpyrazine monosulphate, C6H3(0Me)2,C4H2N2,C6H3(0Me)2,H2S04, which formed orange-red needles. If, however, the solution contained any excess of sulphuric acid, the orange-coloured crystals of the monosulphate soon gave place to small, jet-black prisms of the corre- sponding disulphate, C0H3(OMe)2,C4H2N2-C6H3(OMe)2,2H2SO4. These crystals possessed a brilliant metallic lustre, and were per- manent in the air, although they were dissociated fairly readily by alcohol. A dilute solution of mTO/pp,-tetramethoxy-2: 5-diphenyl- pyrazine disulphate in concentrated sulphuric acid had an intensely blue colour. The amount of these salts was not sufficient for analysis. Salts of mm'pp'-Tetramethoxy-2 : 5-diphenylpyrazine.—This base readily yielded a crystalline monohydrochloride, monohydrobromide, and monosulphate, but no di-acid salt of it could be crystallised. No evidence of the existence of a dihydrochloride could be obtained, but a compound of a bluish-black colour with a bronze lustre, which](https://iiif.wellcomecollection.org/image/b22433181_0014.jp2/full/800%2C/0/default.jpg)