High-frequency currents / by Frederick Finch Strong.
- Strong, Frederick Finch.
- Date:
- [1908], ©1908
Licence: Public Domain Mark
Credit: High-frequency currents / by Frederick Finch Strong. Source: Wellcome Collection.
Provider: This material has been provided by the Harvey Cushing/John Hay Whitney Medical Library at Yale University, through the Medical Heritage Library. The original may be consulted at the Harvey Cushing/John Hay Whitney Medical Library at Yale University.
48/320 (page 22)
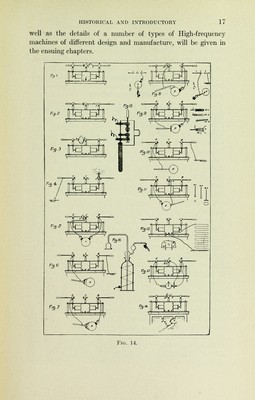
![hours to produce this small quantity of the heaviest of known metals. Whence did this power come? How and why was it directed in this particular channel? Here we are met by the impenetrable secret of creative agency. The sketch (Fig. 15) is intended to give a graphic representa- tion of the break down of a Uranium atom. Some of the freed electrons have already re-formed into Helium atoms; the ma- Fig. 15.—Schematic Diagram Showing Spontaneous Degeneration of Atoms. I.—Uranium Atom (Ur), breaking down into the Radium Atom (Ra), and the Helium Atoms (He). II.—Radium Atom (Ra), breaking down into a Simpler Atom (Bi), (which is probably Bismuth or Lead) and the Helium Atoms (He), giving out meanwhile free Electrons (E, E, 5), (Cathode Rays) and Ether Waves of various frequencies; ],X-Rays; 2, Ultra-Violet Rays; 3, Light Rays; 4, Heat Rays. (Redrawn and Revised from a Sketch by Doctor Batten, in the Archives of the Roentgen Ray.) jority of the remaining electrons ultimately unite to form a single atom which would appear much larger than the Helium atoms but slightly smaller than the original Uranium atom. Heat rays (4), Ultraviolet-light rays (2), X-Rays (1), and streams of free electrons (Cathode Rays, 5) are differ- ent varieties of radiant energy given off in the breaking-down process.](https://iiif.wellcomecollection.org/image/b2100206x_0048.jp2/full/800%2C/0/default.jpg)