Outlines of physical chemistry / by A. Reychler ; translated form the French with the author's permission by John McCrae.
- Reychler, A. (Albert), 1854-
- Date:
- 1904
Licence: In copyright
Credit: Outlines of physical chemistry / by A. Reychler ; translated form the French with the author's permission by John McCrae. Source: Wellcome Collection.
76/302 page 56
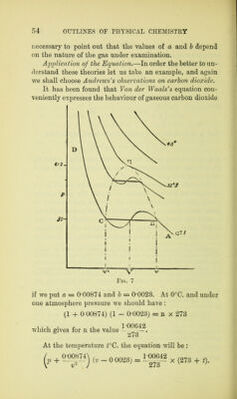
![and from these three equations we can deduce the characteristics of the critical state, vupu t1? expressed in terms of a, 5, and n : the critical volume. • • vx = 3b „ „ pressure . . px = ^5 „ „ temperature . . Tx = For carbon dioxide, the critical temperature thus calculated (82*5°) approximates fairly closely to that experimentally found by Andrews (81°). For the pressure and volume the agreement is not quite so good (61*2 and 0*0069, instead of 74*6 and 0*00447 found experimentally).1 The constants a and b and the critical temperature of other substances have also been established by calculation in quite a satisfactory manner. The Theory of the Corresponding States.—If in Van der Waals's equation we put p = 7rpu v = (f)V] and t = 6 tx, that is to say, if we consider the actual pressure p as a fraction tt of the critical pressure^, and in the same way the volume and temperature as fractions, </> and 0, of the critical volume and temperature ; and if at the same time we express a and b in terms of px and vx : b = a = 27 b3 = 8pxvx2: o then after some simple transformations we obtain (*■ + (3 <p - 1) = 8 d. 1 Van der Waals's equation gives a very good qualitative interpre- tation of the facts observed by Andrews. From the quantitative point of view the interpretation is not quite so good. The reader will gain a good idea of this inexactitude by calculating the values of p, for a given temperature, which correspond to different values of v, and describing the isotherm thus determined beside the experi-](https://iiif.wellcomecollection.org/image/b2813400x_0076.jp2/full/800%2C/0/default.jpg)