Volume 1
A treatise on chemistry / by H.E. Roscoe and C. Schorlemmer.
- Henry Enfield Roscoe
- Date:
- 1877-1892
Licence: Public Domain Mark
Credit: A treatise on chemistry / by H.E. Roscoe and C. Schorlemmer. Source: Wellcome Collection.
600/792 page 584
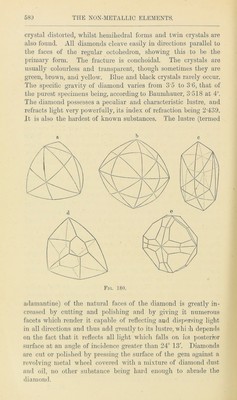
![however, now almost exhausted, not having been worked for the last twenty years. In the sixteenth and seventeenth centuries they were so productive as to yield an annual revenue of £40,000, although they were only worked a few weeks in the year for fear of exhausting the mine. The Keswick Pencil Company still possess, however, a considerable stock of Borrow- dale black-lead, locally called “ wad,” which they use for making the best kind of pencils. Graphite is also found at Passau in Germany, in Bohemia and in Styria. It likewise occurs in many places in the United States, the deposits at Sturbridge, Mass., and at several localities in New York being large enough to yield a considerable supply. By far the largest mine in the United States is the “Eureka Black-Lead Mine” at Sonora in California. The graphite here forms a layer of some twenty to thirty feet in thickness. It is so pure that it may be obtained in large blocks. In the year 1868 not less than one million of kilos, were raised each month.1 Large quantities of graphite are also found in Ceylon. In Southern Siberia this substance occurs in considerable quantities in the Batougal mountains, and is largely exported to Europe. Graphite commonly occurs in compact foliated or granular masses, but occasionally in small six-sided tables, which accord- ing to Kenngott belong to the hexagonal, but according to Nordenskjold to the monoclinic system. It has a steel-grey colour, an unctuous touch, and it is so soft that it gives a black streak on paper. Its specific gravity varies from 2 015 to 2 583, and this considerable variation is due to the fact that almost all natural graphite contains more or less impurity which, when the graphite is burnt, remains behind as ash and consists of alumina, silica, and ferric oxide, with small traces of lime and magnesia.2 It usually contains 0-5 to 1*3 per cent, of hydrogen,3 a fact which seems to point to its organic origin. Graphite is a good conductor of heat and electricity, whilst the diamond is a non-conductor. According to Gustav Pose, the crystalline variety is more difficultly combustible, whereas the compact variety burns more readily than the diamond. The artificial preparation of graphite by melting cast iron containing a large proportion of carbon, and allowing it to cool slowly, was first observed by Scheele in 1778. Cast iron is 1 Chcm. News, 1868, 299. 2 Mene, Comptes JRcndus, Ixiv. 1019 and 1867. 3 Regnault, Ann. Chim. Phys. [2], i. 202.](https://iiif.wellcomecollection.org/image/b28122409_0001_0602.jp2/full/800%2C/0/default.jpg)