Protoplasm : its definition, chemistry and stucture / by Gustav Mann.
- Gustav Mann
- Date:
- 1906
Licence: In copyright
Credit: Protoplasm : its definition, chemistry and stucture / by Gustav Mann. Source: Wellcome Collection.
Provider: This material has been provided by The Royal College of Surgeons of England. The original may be consulted at The Royal College of Surgeons of England.
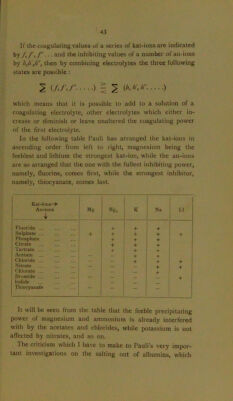
47/62 page 45
![electricity. Diagrammatically an ordinary electrolyte and a colloidal electrolyte (see also p. 28) may be represented in this wa}'. [X.]*- ordinary electrolyte colloidal electrolyte What leads to the formation of colloidal matter ? There is one factor which we may assume to be constant, namely, our solvent water. When water comes into contact with difterent salts, we find that the radicals which go to form the salt, possess the power of becoming ionised to different degrees. The greater the power of a certain radical to become either a kat-ion or an an-ion the greater will also be its influence on other radicals, because its chemical power is in direct ratio to its capacity of becoming ionised. The less its power of form- ing ions the more will it tend to unite with other units similar to or identical with itself, because the least difference of potential must always be developed in a communit}' of iden- tical units. We thus have on the one hand such chemically exceedingly active substances as the halogen- and oxy-acid salts of the alkalies \e.g. KCl, KNO^, K^SOJ, and on the other hand the chemically inactive paraffins [CH^, C^HJ. While the former because of their great ionic power in the presence of water will always interact with other active units and thus lose their individuality, paraffins on the other hand will under the same circumstances remain inactive. It is to me ex- ceedingly interesting to see how our most primitive ancestors, the paraffins, by evolution gave rise to alcohols, aldehydes and acids, and thereby gradually acquired the power of inter- acting with the environment. H H H—C-C-H H H ethane. H H—C—C H OH O acetic acid nh, H-C—C H glycocol). While an ordinary mineral acid such as hydrochloric acid](https://iiif.wellcomecollection.org/image/b22471303_0049.jp2/full/800%2C/0/default.jpg)