A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton.
- Francis Sutton
- Date:
- 1871
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton. Source: Wellcome Collection.
296/452 page 272
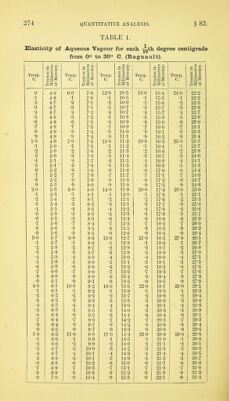
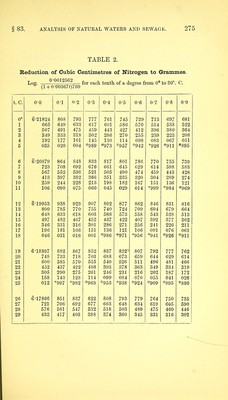
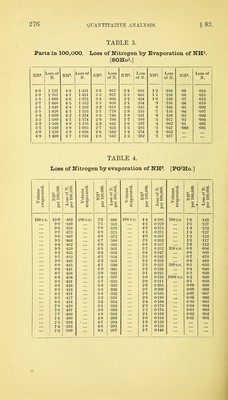
![in which w — the weight of nitrogen, v the volume, p the pressure corrected for tension of aqueous vapour, and t the temperature in degrees centigrade. To facilitate this calculation, there is given in Table 3 the logarithmic value of the expression +^00^67 f) 760 for each tenth of a degree from 0° to 29-9° C, and in Table 4 the tension of aqueous vapour in millimetres of mercury. As the measuring tube is always kept moist with water, the gas when measured is always saturated with vapour. The following example will show the precise mode of calculation:— Total. After absorption of CO2. Nitrogen. Volume of gas, .... 4-4888 c. c. 0-26227 c.c. 0-26227 c.c. Temperature, 13-5° 13-6° 13-7° m.m. Height of mercury in a,c,d, 315-2 485-3 485-3 „ „ ,, b, 198-7 348-8 333-5 Difference, . . . 116-5 136-5 151-8 Plus tension of aqueous vapour at 135° 0, 11-5 11-6 11-7 128-0 Deduct correction for capil- Add for 1 larity, 0-9 capillarity] 2 2 2-2 127-1 150-3 165-7 Deduct this from height 769'8 769-8 769-8 of barometer, .... 127-1 150-3 165-7 Pressure on dry gas, . 642-7 . 619-5 604-1 Log. of volume of gas, . . 0-0012562 0-65213 6-19724 1-41875 6t-19709 1-41875 619694 ” (1 + 0-00367 t) 760 ,, pressure on dry gas, 2-80801 2-79204 2-78111 3-65738 4-40788 4-39680 Log. of weight of gas calcu- lated as hi, . . . . = 0-0045434 0-0002558 0-0002494 gin. From these weights, those of carbon and of nitrogen are obtained by the use of the formulae above mentioned. Thus— A-B = 0-0042876 B + C - 0*0005052 x 3 -_2 -r- 7 )0-0128628 Weight of nitrogen, 0-0002526 Weight of carbon, 0-001837 When carbonic oxide is found, the corresponding weight of nitrogen may be found in a similar manner, and should be added](https://iiif.wellcomecollection.org/image/b28066121_0296.jp2/full/800%2C/0/default.jpg)