A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton.
- Francis Sutton
- Date:
- 1871
Licence: Public Domain Mark
Credit: A systematic handbook of volumetric analysis, or, The quantitative estimation of chemical substances by measure, applied to liquids, solids and gases / by Francis Sutton. Source: Wellcome Collection.
368/452 page 344
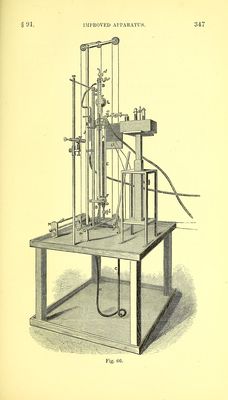
![acid and carbonate in contact, and the increase of volume was due to the carbonr anhydride evolved. The results thus obtained are extremely concordant. In eight experiments with sodic carbonate the percentage of carbonic anhydride found varied from 41-484 to 41*607, theory requiring 41-509. Thirteen experiments with calc spar gave from 43*520 to 43*858, the theoretical percentage being 44*0 ; and in nine other analyses from 43 581 to 43*901 were obtained. Two experiments were made with manganic peroxide, oxalic acid and sulphuric acid, and gave 58*156 and 58*101 per cent, of carbonic anhydride. Some determinations of the purity of magnesium were also per- formed by dissolving the metal in hydrochloric acid and measuring the resulting hydrogen; four operations gave numbers varying between 8*255 and 8*282 ; the metal should yield 8*333. Dr Russell has also employed this process for the determination of the combining proportions of nickel and cobalt (Journ. Chem. Soc. FT. S., vii. 294). MM. Regnault and Reiset described (Ann. Chim. Phys. [3] xxvi. 333) an apparatus by which absorptions could be rapidly con- ducted by means of liquid re-agents brought in contact with the gases in a laboratory tube. The measurements are made in a graduated tube which can be placed in communication with the laboratory tube by means of fine capillary tubes provided with stop cocks, the lower end of the measuring tube being connected by an iron socket and stopcock with another graduated tube in which the pressure, to which the gas is subjected, is measured. The measuring and pressure tubes are surrounded by a cylinder of water. An apparatus similar in principle to this has recently been constructed by Dr Frankland, and is fully described in the section on water analysis, § 83 page 265. Frankland and Ward (Chem. Soc. Jour.- vol. vi. p. 197) made several important improvements in the apparatus of Regnault and Reiset. They introduced a third tube (fig. 65), closed at the top with a stopper, and which is made to act as a barometer to indicate the tension of the gas in the measuring tube, thus rendering the operation entirely independent of variations of atmospheric pressure. The correctionror aqueous vapour is also eliminated by introducing a drop of water into the barometer as well as into the measuring tube, the pressures produced by the aqueous vapour in](https://iiif.wellcomecollection.org/image/b28066121_0368.jp2/full/800%2C/0/default.jpg)