The tautomerism of glyoxalines and the constitution of pilocarpine / by Frank Lee Pyman.
- Pyman, Frank Lee.
- Date:
- [between 1910 and 1919?]
Licence: In copyright
Credit: The tautomerism of glyoxalines and the constitution of pilocarpine / by Frank Lee Pyman. Source: Wellcome Collection.
2/44 page 1815
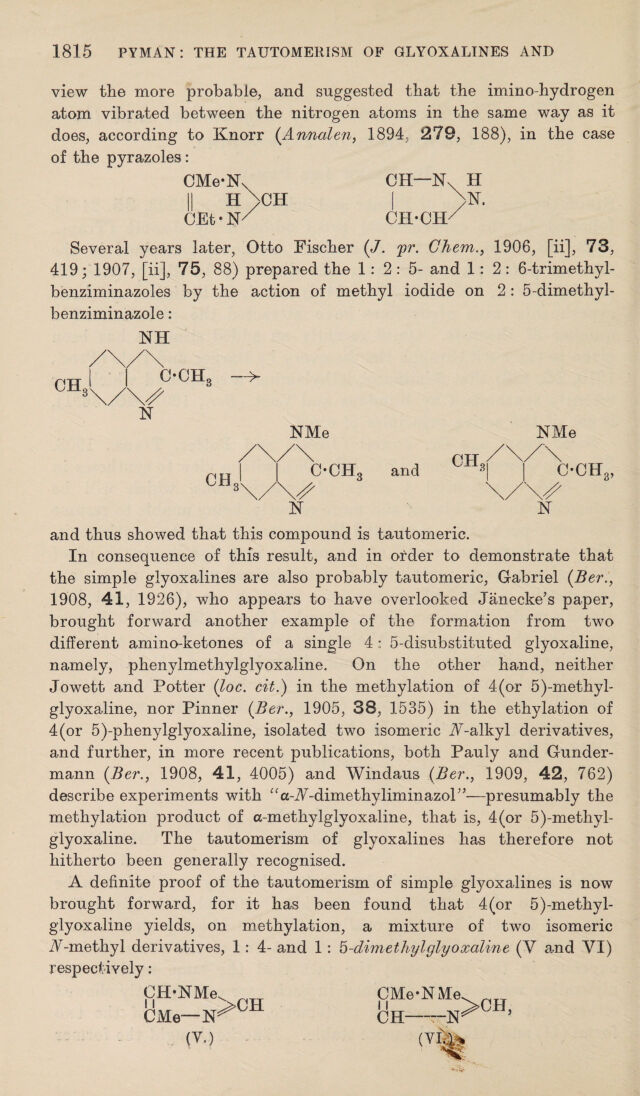
![view the more probable, and suggested that the imino-hydrogen atom vibrated between the nitrogen atoms in the same way as it does, according to Knorr {Annalen, 1894, 279, 188), in the case of the pyrazoles: CMe-N. CH—N. H II H>CH I >h[. CEt-N/ CH-CH/ Several years later, Otto Fischer (/. pr. Chem.^ 1906, [ii], 73, 419; 1907, [ii], 75, 88) prepared the 1: 2 : 5- and 1:2: 6-trimethyl- benziminazoles by the action of methyl iodide on 2 : 5-dimethyl- benziminazole: NH and thus showed that this compound is tautomeric. In consequence of this result, and in order to demonstrate that the simple glyoxalines are also probably tautomeric, Gabriel 1908, 41, 1926), who appears to have overlooked Janecke’s paper, brought forward another example of the formation from two different amino-ketones of a single 4: 5-disubstituted glyoxaline, namely, phenylmethylglyoxaline. On the other hand, neither Jowett and Potter {loc. cit.) in the methylation of 4(or 5)-methyl- glyoxaline, nor Pinner {Ber., 1905, 38, 1535) in the ethylation of 4(or 5)-phenylglyoxaline, isolated two isomeric iV-alkyl derivatives, and further, in more recent publications, both Pauly and Gunder- mann (Ber., 1908, 41, 4005) and Windaus {Ber., 1909, 42, 762) describe experiments with ^^a-i\^-dimethyliminazoP’—presumably the methylation product of a-methylglyoxaline, that is, 4 (or 5)-methyl- glyoxaline. The tautomerism of glyoxalines has therefore not hitherto been generally recognised. A definite proof of the tautomerism of simple glyoxalines is now brought forward, for it has been found that 4(or 5)-methyl- glyoxaline yields, on methylation, a mixture of two isomeric A-methyl derivatives, 1: 4- and 1 : b-dim ethyl glyoxaline (V and VI) respectively: CH-NMe CMe—N >CH . (V.) CMe-NMe 11 CH—-N >0H,](https://iiif.wellcomecollection.org/image/b30622220_0002.jp2/full/800%2C/0/default.jpg)