The elements of bacteriological technique : a laboratory guide for medical, dental, and technical students / by J.W.H. Eyre.
- Eyre, John W. H. (John William Henry), 1869-
- Date:
- 1913
Licence: In copyright
Credit: The elements of bacteriological technique : a laboratory guide for medical, dental, and technical students / by J.W.H. Eyre. Source: Wellcome Collection.
519/564
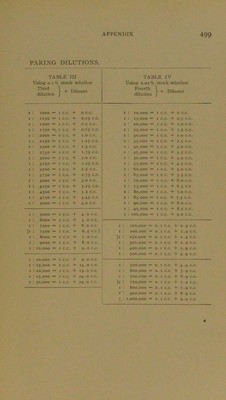
![From dilutions of i: 10,000 to i; 100,000 rise by increments of 5000. From dilutions of i: 100,000 to 1; 1,000,000 rise by increments of 100,000. When dealing with a substance of unknown powers—and this is especially true with regard to agglutinating sera—it is customary to run a preliminary test, using a few widely separated dilutions such as may be obtained in the following manner; First Dilution—I. I c.c. serum + g c.c. normal saline solution = 10 per cent, solution or 1:10 dilution (of which i c.c. contains o.i c.c. of the original serum). When dealing with fluids other than serum the diluent is usually distilled water; whilst if the original substance is a solid the instructions would read: I gram o.s. + 10 c.c. distilled water = 10 per cent, solution, etc. Secon-d Dilution—II. I c.c. fir.st dilution + g c.c. normal saline solution = i per cent, solution or i: 100 dilution. Third Dilution—III. I c.c. second dilution + 9 c.c. normal saline solution == i per millc solution or i; 1000 dilution. Fourth Dilution—IV. I c.c. second dilution + 9 c.c. normal saline solution = 0.1 ]>er mille solution or i : 10,000 dilution. The following tables showing the secondary dilutions that can readily be prepared from each of these four primary dilutions for use in the subsequent determination of the exact litre will probably be found of service by those who are not ready mathematicians. 32](https://iiif.wellcomecollection.org/image/b28108255_0519.jp2/full/800%2C/0/default.jpg)