Adolph Strecker's Short text-book of organic chemistry / by Dr. Johannes Wislicenus .. ; translated and edited with extensive additions, by W.R. Hodgkinson ... and A.J. Greenaway.
- Strecker, Adolph, 1822-1871.
- Date:
- 1881
Licence: Public Domain Mark
Credit: Adolph Strecker's Short text-book of organic chemistry / by Dr. Johannes Wislicenus .. ; translated and edited with extensive additions, by W.R. Hodgkinson ... and A.J. Greenaway. Source: Wellcome Collection.
43/874 page 19
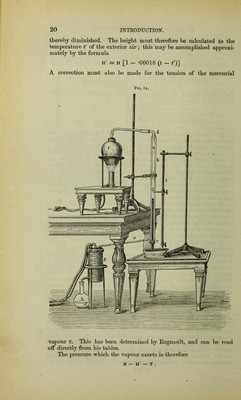
![balloon at the temperature t' is, if h be the coefficient of cubic expansion of the glass (free from lead = *00255)— v' = v (1 + h t'). Therefore the weight of an equal volume of air under like cir- cumstances = *0012932 .v [1 +**']• l + -00367*' * 760 the vapour density d of the body being (without correction for any residual air in the balloon) D= p'-(p-^) •0012932 . y . (1 + kt’) . x + .00367*' ‘ 760 29. Two important methods based upon the second principle are now in use—viz. the methods of Hoffmann and Victor Meyer, which are based upon the method of Gay-Lussac, now seldom used. Hoffmann’s method is applicable to bodies whose boiling point is considerably below 100° C., and whose vapours already follow the laws of Marriotte and Gay-Lussac at the boiling point of water. This excellent method may also be used for the vapour density de- termination of less volatile bodies, as by the employment of a torri- cellian vacuum accurate measurements of the volumes may be made at a much lower temperature than the body’s actual boiling point under atmospheric pressure. A graduated barometer tube about 1 metre long a (fig. 14) is completely filled with dry mercury and inverted in a vessel full of mercury. The tube is then surrounded by a wider tube b, fitted tightly to it by the cork c. At the upper end of this surrounding tube a tube d is fitted for the entrance of vapour, whilst an exit tube, united to a condenser, is attached to its lower end. A small bottle of 1 cc., or less, content, provided with a glass stopper, is completely filled with the substance under investigation, and the weight p of the contents determined by weighing. The bottle is then sent up the barometer tube,' when the stopper is generally expelled by the excess of interior pressure. A good stream of the vapour of some substance of known boiling point is then passed through the annular space between the cylinder and measuring tube, by which the latter and its contents are soon raised to a like tempe- rature. The substances generally used in the vapour bath are water for the more volatile and aniline for the less volatile bodies. The liquid contained in the bottle is converted into vapour, de- pressing the mercurial column. As soon as this has reached a constant position in the measuring tube the volume of vapour v is read off, together with the atmospheric barometric pressure b, the height h of the mercurial column inside the measuring tube and the boiling point of the liquid used for the vapour bath being also noted. When aniline or other high-boiling liquids have been employed, the pressure on the vapour at the temperature t is not simply = b — h, the interior mercurial column being considerably heated and its sp. gr.](https://iiif.wellcomecollection.org/image/b28057454_0043.jp2/full/800%2C/0/default.jpg)