Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman.
- Ettore Molinari
- Date:
- 1912
Licence: In copyright
Credit: Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman. Source: Wellcome Collection.
47/734
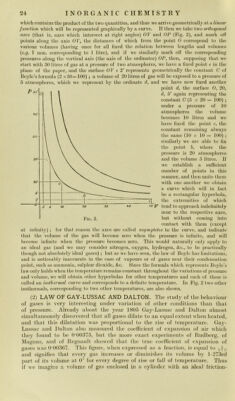
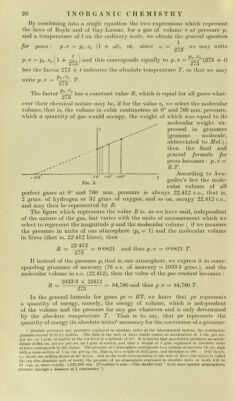
![Jess and weiglitJess piston, at 0°, and il we divide ilie cyiinder into 27.‘i part.s, then Oli ]o\\ering llie temperature to 273° below zero thè gas will diminisli in volume by of its originai volume (at 0°) for eacli degree and will not finally occiipy any volume wliatever, tliat is, will bc deprived of every forni of energ}^ or niatter, ivliicli is absnrd ; we iiiay say more correctly tliat below tliis temperature thè energy of its material will reniain Constant. (If tem- peratures below absolute zero bave any meaning.—Translator’’s note.) This liniiting temperature, with regard to which thè common zero point of ther- nionieters would be + 273°, is callcd thè absolute zero. Thus at 272° below zero a gas would really occupy of its volume at 0°. On raising thè tem- perature thè volume will constantly increase and at 273° above absolute zero, tliat is, at thè ordinary zero temperature, it will occupy its originai volume, which is 273 times larger than thè volume which it occupied at 273° below zero. Thus thè volume of a gas is proportional to its absolute temperature, and thus at 273° above thè ordinary zero point a gas will acquire doublé thè volume which it occupies at thè ordinary zero, or 546 times its volume at - 272°. The conception of absolute temperature is also iiiteresting because it enables one to understaiid why it is iiiipossible to completely transform thermal into meclianical energy. The actual mechanical work which can be produced by thè interchange of heat betwecn two substances is proportional to thè temperature difference tlu:ough which thè transformation of heat into work occurs. The greater thè difference in temperature between thè two sub- stances thè greater will be thè yield of mechanical work ; but it follows that in order to transform all thè heat in a body into mechanical work it would be necessary to lower its temperature to absolute zero, which is practically impossible (see also below, and p. 7, “ Degradation of Energy”)* The law of Gay-Lussac and Dalton may also be represented by a mathematica! formula in which V indicates thè volume of a gas at thè temperature T, and V' thè volume of thè same quantity of gas by weight at thè temperatme T' ; thè volumes being proportional to thè absolute temperatures we ha ve : V T' V : V'^T: T', or V'.T^ V.T', that is, V'= -y-. If, for example, a gas occupies a volume F of 4 litres at thè temperature, T, of 300° absolute, that is, at 27° above thè ordinary thermometric zero, it will occupy a volume F' of 8 litres at thè temperature, T', of 600° absolute, that is, 327° above thè ordinary zero ; controlling this by thè above formula we bave : 4 X 600 = 8 X 300 V.T' = V'.T If we denote thè coefficient of dilatation of a gas by a, and its volume measured at thè temperature t° of thè ordinary scale by v, then thè volume of a gas at 0° will be Vq, and at t° it will.be vt ; then we havc thè equation : vt^v^y (1 + ut), ahvays under thè condition that thè pressure remains Constant. If we takc thè volume at 0° as l,.then at 10° it will be 1-0367, at 20° i;^=T073 at 50° 1-184, and at 100° 1-367, &c. The graphic representation will be a curve of thè first degree, sincc thè two variables are always present in thè first power and are not nuiltiplied by one another, and will lead to a linear function, so that thè law of Gay-Lussac and Dalton will be represented by a straight line. If we actually talee thè temperatures t as abscissìe and thè volumes v as ordinatcs (Fig. 3) then on drawing a curve through thè points corresponding to thè various pressures and corresponding volumes we obtain a straight linC; which, if prolonged in thè direction of thè temperatures below 0° shows a zero volume at —273°. We bave seen that thè formula of Gay-Luswac’s law applics if thè pressure of thè gas remains Constant during heating ; if, on thè other hand, thè volume of thè gas is kepi Constant vvhilst it is heated its pressure will necessarily rise and then thè pressure pi at t°, expressed as a function of thè pressure Po at 0°, will be Pi — pQ {\ -j- at). I](https://iiif.wellcomecollection.org/image/b28134187_0049.jp2/full/800%2C/0/default.jpg)