Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman.
- Ettore Molinari
- Date:
- 1912
Licence: In copyright
Credit: Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman. Source: Wellcome Collection.
50/734
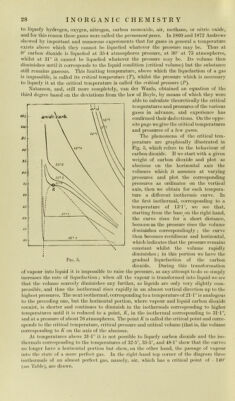
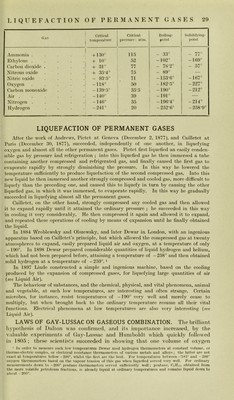
![to liquefy hydrogen, oxygcn, nitrogeii, carbon moiioxido, air, nicthane, or nitric oxidc, and for tliis reasoii thesc gases wcrc called tlie 'permanent gases. In 1869 and 1872 Andrews slioAvcd by important and numerous cxpcriments that for gases in generai a temperature cxists above Avliich they eannot be liquelied wliatever thè pressure may be. Thus at 0° earbon dioxidc is liqueficd at 35-4 atmospheres pressure, at 30° at 73 atinospheres, wliilst at 31° it eannot be liquefìed whatever thè pressure may be. Its volume then diminishes until it eorresponds to thè liquid condition (critieal volume) but thè substance stili remains gaseous. This liniiting temperature, above which thè liquefaction of a gas is impossiblc, is called its criticai temperature {T), whilst thè pressure which is necessary to liquefy it at thè criticai temperature is called thè criticai pressure (P). Natanson, and, stili more completely, van der Waals, obtained an cquation of thè third degree based on thè deviations from thè law of Boyle, by means of which they were able to calculate theoretically thè criticai tem^icratures and pressures of thè various gases in advance, and expcrience has confirmed their deduetions. On thè oppo- site i^age we givo thè criticai temj)eratures and pressures of a few gases. The phenomena of thè criticai tem- perature are graphically illustrated in Fig. 5, which refers to thè behaviour of carbon dioxide. If we start with a given weight of carbon dioxide and plot as abscissa3 on thè horizontal axis thè volumes which it assumes at varying pressures and plot thè corresponding pressures as ordinates on thè vertical axis, then we obtain for each tempera- ture a different isothermic curve. In thè fìrst isothermal, corresponding to a temperatiue of 13T°, we see that, starting from thè base on thè right hand, thè curve rises for a short distance, because as thè pressure rises thè volume diminishes correspondingly ; thè curve then becomes rectilinear and horizontal, which indicates that thè pressiue remains Constant whilst thè volume rapidly diminishes ; in tliis ])ortion we have thè graduai liquefaction of thè carbon dioxide. During this transformation of vapour into liquid it is impossible to raise thè pressure, as any attempt to do so simply increases thè rate of liquefaction ; when all thè vapour is transformed into liquid we see that thè volume scarcely diminishes any further, as liquids are only very slightly coni- pressiblc, and thus thè isothermal rises rapidly in an almost vertical direction up to thè highest pressures. The next isothermal, corresponding to a temperature of 21 T° is analogous to thè precedi ng one, but thè horizontal porti on, wherc vapoiu and liquid earbon dioxide coexist, is shorter and continues to diminish in thè isothermals corresponding to higher temperatures until it is redueed to a point, K, in thè isothermal corresponding to 31 T°, and at a pressure of about 76 atmospheres. The point K is called thè criticai point and corrc- sponds to thè criticai tempcratm-e, criticai pressiuc and criticai volume (thatis, the volume corresponding to K on the axis of the abscisspe. At temperatures above 31 T° it is not possiblc to liquefy carbon dioxide and thè iso- therjuals corres|)onding to the temperatures of 32*5°, 35-5°, and 48T° show that the curves no loiiger have a horizontal portion but show, on the other hand, thè passage of vapour iiit(j the state of a more perfect gas. Iji the right-hand to[) corner of the diagram three isothermals of an almost perfect gas, namely, air, which has a criticai point of — 140° {see Table), are diawn.](https://iiif.wellcomecollection.org/image/b28134187_0052.jp2/full/800%2C/0/default.jpg)