Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman.
- Ettore Molinari
- Date:
- 1912
Licence: In copyright
Credit: Treatise on general and industrial inorganic chemistry / by Etore Molinari ; third revised and amplified Italian edition translated by Ernest Feilman. Source: Wellcome Collection.
51/734
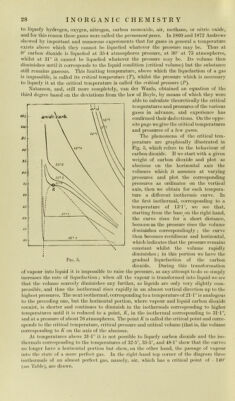
![Gas Criticai temperature Criticai pressure: atni. Boiling- point Solidifying- poitit Ammoni a . . . . . + 130° 115 - 33° - 77° Ethylene . . . . . + 10° 52 -102° -169° Carlicm dioxidc .... + 31° 77 - 78-2° - 57° Nitrous oxido .... + 35*4° 75 - 89° — Nitric oxiclc .... - 93-5° 71 -153-6° -167° Oxygen . . . . . -118° 50 - 182-5° -227° Carbon monoxide -139-5° 35-5 -190° -212° Air ...... -140° 39 -191° — Nitrogen . . . . . -146° 35 -196-4° -214° Hydrogen . . . . -241° 20 -252-6° -258-9° LIQUEFACTION OF PERMANENT GASES After thè work of Andrews, Pictet at Geneva (Docember 2, 1877), and Cailletet at Paris (December 30, 1877), siicceeded, independently of onc another, in liquefying oxygen and almost all thè other pcrmanent gases. Pictet first licj[ueficd an easily conden- sible gas by pressure and refrigeration ; into this liquefied gas he thcn immcrsed a tube containing another compresseci and refrigerated gas, and finally caused thè first gas to evaporate rapidly by strongly diminishing thè pressure. In this way he lowered thè temperature sufficiently to produco liquefaction of thè second compresseci gas. Into this new liquicl he then immerseci another strongly compressed and cooled gas, more clifficult to liquefy than thè prececling one, and caused this to liquefy in turn by eausing thè other liquefied gas, in which it was immersed, to evaporate rapidly. In this way he gradiially succeeded in liquefying almost all thè permanent gases. Cailletet, on thè other hand, strongly compressed any cooled gas and then allowed it to expancl rapidly until it attained thè ordinary pressure ; he succeeded in this way in cooling it very consiclerably. He then compressed it again and allowed it to expand, and repeated these operations of cooling by means of expansion until he finally obtained thè liquicl. In 1884 Wroblewsky and Olszewsky, and later Dewar ia London, with an ingenious apparatus based on Cailletet’s principio, but which allowed thè compresseci gas at twenty atmospheres to expand, easily prepared liquicl air and oxygen, at a temperature of only -190°. In 1898 Dewar prepared considerable quantities of liquid hydrogen and helium, which had not been prepared before, attaining a temperature of -258° and then obtained solid hydrogen at a temperature of - 259°. i In 1897 Linde construeted a simple and ingenious machine, based on thè cooling produced by thè expansion of compressed gases, for liquefying largo quantities of air {see Liquicl Air). The behaviour of substances, and thè Chemical, physical, and vi tal phenomena, animai and vegetable, at sudi low temperatures, are interesting and often strange. Certain microbes, for instance, resist temperatures of -190° very woll and merely cease to multiply, but when brought back to thè ordinary temperature résumé all thoir vital functions. Electrical phenomena at low temperatures are also very interesting {see Liquicl Air). LA WS OF GAY-LUSSAC ON GASEOUS COMBINATION. Thebrilliant hy])othesis of Dalton was confirmecl, and its importance increased, by thè valiiable experiments of Gay-Lussac and Humboldt which quickly folloued in 1805 ; these scientists succeeded in showing that one volume of oxygen ‘ In order to measure sudi low temperatures Dewar uscd hydrogen tlicrmometers at Constant volume, or thermo-electric couples, or electrical rcsistance thermometers of various mctals and alloys ; thè latter are not cxact at temperatures below -200°, wliilst thè first are thè hest. For temperatures between -'183° and -200° oxygen thermometers based on thè vapour tension of this gas when liquefied served very well. For ordinary iiieasuremeiits down to -200° iientane thermometers served sulliciently well; iientane, Or,fI|-_>. obtained from thè more volatile petrolcum fractions, is already liquid at ordinary temperatures and remains liquid down to about -200°.](https://iiif.wellcomecollection.org/image/b28134187_0053.jp2/full/800%2C/0/default.jpg)