Manual of chemical analysis as applied to the examination of medicinal chemicals : a guide for the determination of their identity and quality, and for the determination of their identity and quality, and for the detection of impurities and adulterations : for the use of pharmaceutists, physicians, druggists, and manufacturing chemists, and of pharmaceutical and medical students / by Frederick Hoffmann.
- Friedrich Hoffmann
- Date:
- 1873
Licence: Public Domain Mark
Credit: Manual of chemical analysis as applied to the examination of medicinal chemicals : a guide for the determination of their identity and quality, and for the determination of their identity and quality, and for the detection of impurities and adulterations : for the use of pharmaceutists, physicians, druggists, and manufacturing chemists, and of pharmaceutical and medical students / by Frederick Hoffmann. Source: Wellcome Collection.
Provider: This material has been provided by the Francis A. Countway Library of Medicine, through the Medical Heritage Library. The original may be consulted at the Francis A. Countway Library of Medicine, Harvard Medical School.
122/410 page 116
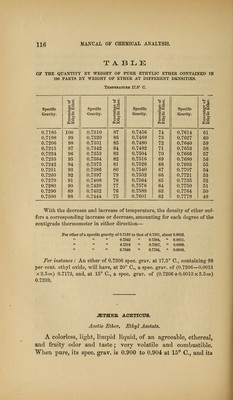
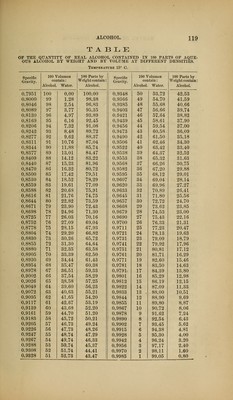
![TABLE OF THE QUANTITY BY WEIGHT OP PURE ETHYLIO ETHER CONTAINED TN 100 PARTS BY WEIGHT OF ETHER AT DIFFERENT DENSITIES. TEMPEKATtTRE 1T.5° C. o «> o « O <w p-3 Specific ^'i Specific t« Specific t« Specific si Gravity. Gravity. Gravity. a ^ Gravity. o >> s-xi 0.7185 100 0.7310 87 0.7456 74 0.7614 61 0.7198 99 0.7320 86 0.7468 73 0.7627 60 0.7206 98 0.7331 85 0.7480 72 0.7640 59 0.7215 97 0.7342 84 0.7492 71 0.7653 58 0.7224 96 0.7353 83 0.7504 70 0.7666 57 0.7233 95 0.7364 82 0.7516 69 0.7680 56 0.7242 94 0.7375 8] 0.7528 68 0.7693 55 0.7251 93 0.7386 80 0.7540 67 0.7707 54 0.7260 92 0.7397 79 0.7552 66 0.7721 53 0.7270 91 0.7408 78 0.7564 65 0.7735 52 0.7280 90 0.7420 77 0.7576 64 0.7750 51 0.7290 89 0.7432 76 0.7588 63 0.7764 50 0.7300 88 0.7444 75 0.7601 62 0.7778 49 With the decrease and increase of temperature, the density of ether suf- fers a corresponding increase or decrease, amounting for each degree of the centigrade thermometer in either direction— For ether of a specific gravity of 0.7198 to that of 0.7331, about 0.0013. 0.7342 0.7504, 0.0011. 0.7516 0.7627, 0.0009. 0.7640 0.7764, 0.0008. For instance: An ether of 0.7206 spec. grav. at 17.5° C, containing 98 per cent, ethyl oxide, will have, at 20° C, a spec. grav. of (0.7206—0.0013 x2.5=) 0.7173, and, at 15° C, a spec. grav. of (0.7206 + 0.0013x2.5=) 0.7239. -aSTHER ACETICUS. Acetic Ether, Ethyl Acetate, A colorless, liglit, limpid liquid, of an agreeal)le, ethereal, and fruity odor and taste; very volatile and combustible. When pure, its spec. grav. is 0.900 to 0.904 at 15^ C, and its](https://iiif.wellcomecollection.org/image/b21059196_0122.jp2/full/800%2C/0/default.jpg)