Meat, milk, and wheat : an elementary introduction to the chemistry of farming : to which is added a review of the questions at issue between Mr. Lawes and Baron Liebig / by Thomas Dyke Acland, jun.
- Acland, Sir Thomas Dyke, 1809-1898.
- Date:
- 1857
Licence: Public Domain Mark
Credit: Meat, milk, and wheat : an elementary introduction to the chemistry of farming : to which is added a review of the questions at issue between Mr. Lawes and Baron Liebig / by Thomas Dyke Acland, jun. Source: Wellcome Collection.
Provider: This material has been provided by The Royal College of Surgeons of England. The original may be consulted at The Royal College of Surgeons of England.
23/104 page 17
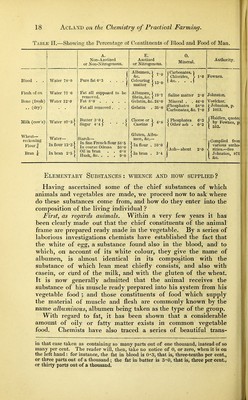
![there combined, and having a strong tendency to pass into the state of ammonia, as soon as vital force ceases, ammonia is rapidly formed and flies off as a gas ; the most offensive smell of putrefying substances is owing to another gas formed with sul- phur, and well known as the smell of rotten eggs.—See Table II., column E. iv. Some parts of Animals and Vegetables contain Earthy Matters.—In addition to the elements which we have found [viz. water, carbon, and nitrogen], the animal system always contains two elements, well known as sulphur and phosphorus ; these are to be found in those parts of an animal which are in the column E, and also in the curd of milk and the gluten of wheat. If we carry the action of heat one step farther, and burn the meat and wheat in the open air, they will gradually consume away till nothing is left except a small amount of ashes, and in these ashes we shall find, in addition to the elements already named, viz. Phosphorus and Sulphur, which form powerful acids, the following earthy or mineral matters :— 1. Potash and soda, commonly called alkalies. 2. Lime and magnesia, called alkaline earths. 3. Iron, which is always present in the blood.* These bodies enter into combination with various acids, of which they are the corresponding bases : as for instance, gypsum consists of a combination of sulphuric acid and lime, and is therefore called sulphate of lime. A large part of our bones consists of phosphoric acid in combination with lime and mag- nesia, called therefore phosphate of lime and phosphate of magnesia.—See Table II., column O. These ashes of mineral and earthy matter may appear insigni- ficant, but, small as they are, in proportion to the bulk of the bodies in which they are found, they are of the highest import- ance. A great part of the theory and practice of manuring depends upon them. Certain very small quantities of them are essential to the health of animals and plants; and however abundantly other fertilizers may be supplied, if any one mineral element which the plants or the animals require is inadequately supplied, they sicken and die. The proportions of the different component parts of blood, and also of meat, milk, and wheat, classified under the four heads just enumerated, are given in the following Table :t— * This list does not profess scientific accuracy or completeness. Common salt, for instance, consists of chlorine and sodium ; and there are other chlorides, but it seemed better here to speak only of familiar substances entering into composi- tion with well-known acids. t As it is possible that some of my readers may have forgotten their decimal fractions, this table and the next are so arranged, that by disregarding the decimal point altogether the figui-es may be read as whole numbers, each quantity being](https://iiif.wellcomecollection.org/image/b22349893_0025.jp2/full/800%2C/0/default.jpg)