Chemical structure and antigenic specificity : a comparison of the crystalline egg-albumins of the hen and the duck / by Henry Drysdale Dakin and Henry Hallett Dale.
- Drysdale Dakin, Henry.
- Date:
- 1919
Licence: Public Domain Mark
Credit: Chemical structure and antigenic specificity : a comparison of the crystalline egg-albumins of the hen and the duck / by Henry Drysdale Dakin and Henry Hallett Dale. Source: Wellcome Collection.
7/16
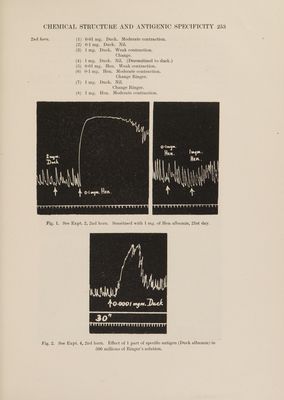
![albumin showed a value of — 9-9°. Pure J-proline has a specific rotation in water of — 77-4°, and while much racemisation ordinarily occurs in the process of isolation, the above values are much lower than those previously observed even from other racemised proteins, so that it appears that most of the proline groups in both proteins were inactivated by the action of alkali. (5) No perfectly pure specimens of tyrosine were obtained, since the more soluble inactive acid in small amounts is hard to separate from leucine and other impurities. No laevo-rotation was observed in any case. (6) The whole of the aspartic acid from the duck egg albumin was optically inactive as well as the mother-liquors from which aspartic acid was separated, so that it is certain that none of the active acid was present. From the hen egg albumin, in addition to a considerable amount of racemic aspartic acid, a small amount (0-3 g.) of the pure laevo-acid was separated. It decomposed around 270°, was faintly laevo-rotatory in alkaline solution, and had [a |29 = + 24-5° in hydrgchloric acid solution (3 mols). Titration with standard alkali using litmus as indicator proved the purity of the product and freedom from leucine. Optically pure aspartic acid under similar conditions shows a specific rotation of + 25-7°. Since in any case /-aspartic acid is largely racemised during its separation by the ester method, the detection of a little of the laevo-acid must be’ taken to indicate the original presence of con- siderably more of the active acid. (7) The whole of the histidine fraction from the hen egg albumin was completely inactive both as free base and as salts. On the other hand the histidine from the duck egg albumin, though small in quantity, was strongly laevo-rotatory, having [a]*2 = -- 27-2°, compared with — 39° for the optically pure base. 70 per cent. of the histidine was therefore made up of the laevo- variety. In each case the histidine was successively separated and identified as phosphotungstate, silver salt, precipitation with mercuric sulphate, and picrolonate. | . (8) In neither case could any dextro-lysine be detected, but it 1s note- worthy that in each case the lysine fraction was feebly laevo-rotatory. The cause of this is obscure, but it was certainly not due to contamination with proline. ANAPHYLACTIC REACTIONS. The experiments were all made on guinea-pigs, each of which received a preparatory injection of 1 mg. of one or other of the albumins. After intervals varying from 19 to 31 days they were tested for sensitiveness. Most of the tests were made, by the now familiar method, on the isolated uterine muscle, young virgin females being chosen for this purpose. The use of this method enabled sensitiveness to both albumins to be tested on the same animal. In most cases some degree of sensitiveness to both was present. In many a high degree of sensitiveness to the albumin not used for the preparatory injection was observed. In all cases, however, a preferential sensitisation](https://iiif.wellcomecollection.org/image/b33454280_0007.jp2/full/800%2C/0/default.jpg)